 
Some other groups in the periodic table display fewer similarities and/or vertical trends (for example Groups 14 and 15), and these have no trivial names and are referred to simply by their group numbers. the alkali metals, alkaline earth metals, halogens and noble gases. In some groups, the elements have very similar properties and exhibit a clear trend in properties down the group - these groups tend to be given trivial (unsystematic) names, e.g. Groups are considered the most important method of classifying the elements. A group is a vertical column in the periodic table of the elements.It should be noted that the properties vary differently when moving vertically along the columns of the table, than when moving horizontally along the rows.  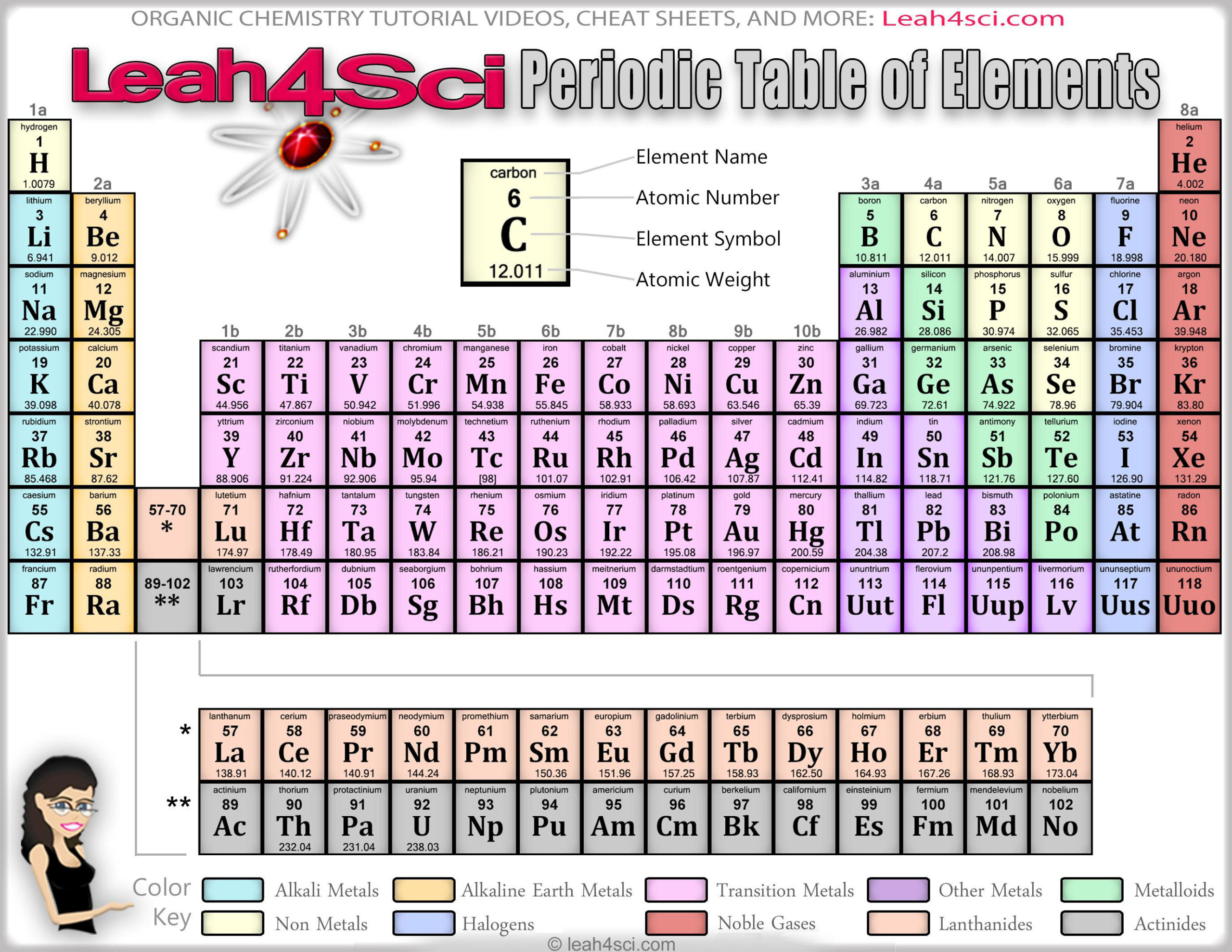
The main value of the periodic table is the ability to predict the chemical properties of an element based on its location on the table. Elements 43 (technetium) and 61 (promethium), although of lower atomic number than the naturally occurring element 92, uranium, are synthetic elements 93 (neptunium) and 94 (plutonium) are listed with the synthetic elements, but have been found in trace amounts on earth. Ninety-two are found naturally on Earth, and the rest are synthetic elements that have been produced artificially in particle accelerators. In printed tables, each element is usually listed with its element symbol and atomic number many versions of the table also list the element's atomic mass and other information, such as its abbreviated electron configuration, electronegativity and most common valence numbers.Īs of 2006, the table contains 117 chemical elements whose discoveries have been confirmed. There are progressively longer periods further down the table, grouping the elements into s-, p-, d- and f-blocks to reflect their electron configuration. According to quantum mechanical theories of electron configuration within atoms, each horizontal row ( "period") in the table corresponded to the filling of a quantum shell of electrons. Rows are arranged so that elements with similar properties fall into the same vertical columns ( "groups"). the number of protons in the atomic nucleus). Elements are listed in order of increasing atomic number (i.e. The layout of the periodic table demonstrates recurring ("periodic") chemical properties. The valences are shaded instead of series.The blocks are shaded instead of series.The wide table sets inline the f-block of lanthanides and actinides.The Electronegativity table provides electronegativities.The detailed table provides a smaller version of the huge table.The huge table provides the above and atomic masses.The big table provides the basics and full element names.A vertical table scrolls down for narrow pages.The standard table (same as above) provides the basics.The Wide Periodic Table incorporates the f-block the Extended Periodic Table incorporates the f-block and adds the theoretical g-block.Įlement categories in the periodic table MetalsĪtomic number colors show state at standard temperature and pressure (0 ☌ and 1 atm) Solidsīorders show natural occurrence PrimordialĪlternative versions ( Layout/view of the table) This common arrangement of the periodic table separates the lanthanides and actinides from other elements. Methods for displaying the periodic table Standard periodic table Group → 1.2 Alternative versions (Layout/view of the table).1 Methods for displaying the periodic table. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
